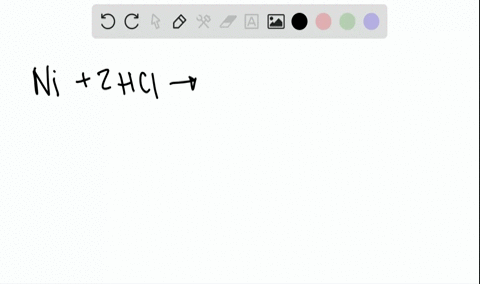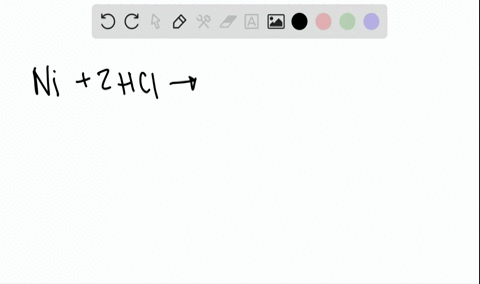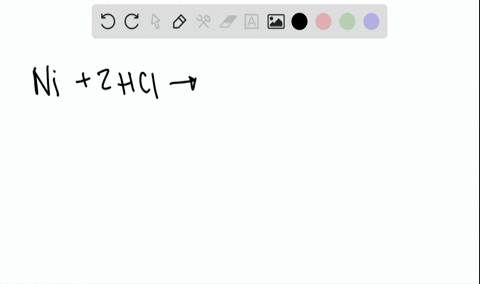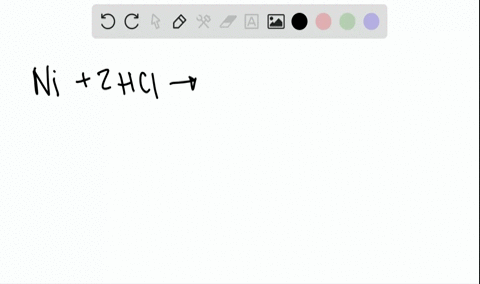
![A given nitrogen containing aromatic compound A reacts with \\[{\\text{Sn\/ HCl}}\\], followed by \\[{\\text{HN}}{{\\text{O}}_{\\text{2}}}\\] to give an unstable compound B. B, on treatment with phenol, forms a beautiful coloured compound C with molecular A given nitrogen containing aromatic compound A reacts with \\[{\\text{Sn\/ HCl}}\\], followed by \\[{\\text{HN}}{{\\text{O}}_{\\text{2}}}\\] to give an unstable compound B. B, on treatment with phenol, forms a beautiful coloured compound C with molecular](https://www.vedantu.com/question-sets/b565ff56-4b65-4f02-a304-a282cc9558db4662046607693484074.png)
A given nitrogen containing aromatic compound A reacts with \\[{\\text{Sn\/ HCl}}\\], followed by \\[{\\text{HN}}{{\\text{O}}_{\\text{2}}}\\] to give an unstable compound B. B, on treatment with phenol, forms a beautiful coloured compound C with molecular

Structure characterization of the Ni–C catalysts. (a) XRD patterns of... | Download Scientific Diagram

Corrosion rates of the Cu–Hf–Ti–Ni–Nb BMGs in 1 N HCl and 3 mass% NaCl... | Download Scientific Diagram

a) Draw the mechanism for the reaction of tin/HCl with m-nitroacetophenone. b) Why is sodium hydroxide added to the reaction? What tin compounds are produced? How do you separate the tin salts

Electrochemical Preparation and Post-treatment of Composite Porous Foam NiZn Alloy Electrodes with High Activity for Hydrogen Evolution | Scientific Reports

a) Draw the mechanism for the reaction of tin/HCl with m-nitroacetophenone. b) Why is sodium hydroxide added to the reaction? What tin compounds are produced? How do you separate the tin salts

Effect of HCl concentration on desorption of Ni(II) from DHHC (amount... | Download Scientific Diagram